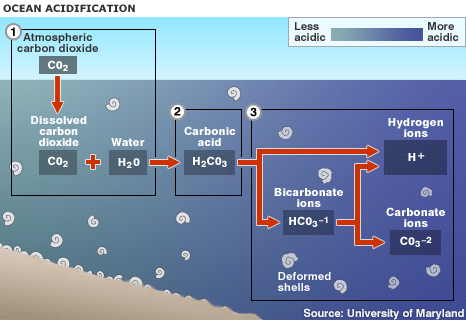
The Chemistry
When carbon dioxide gas (CO2) is absorbed into the ocean, it bonds with water molecules (H2O) producing carbonic acid (H2CO3 (aq)).
CO2 + H2O yields H2CO3
This is obviously acidic when it is dissolved in water.
H2CO3 yields H + HCO3-
Oceans are usually very saturated with carbonate ions (CO3-²), that bond with these extra hydrogen ions to form another bicarbonate ion, now neutralizing the free hydrogen.
H + CO3-² yields HCO3-
This causes the carbonate levels in seawater to decrease, leeching out carbonate ions from calcium carbonate minerals that are essential to the growth of the shells and skeletons of many marine organisms.
CO2 + H2O yields H2CO3
This is obviously acidic when it is dissolved in water.
H2CO3 yields H + HCO3-
Oceans are usually very saturated with carbonate ions (CO3-²), that bond with these extra hydrogen ions to form another bicarbonate ion, now neutralizing the free hydrogen.
H + CO3-² yields HCO3-
This causes the carbonate levels in seawater to decrease, leeching out carbonate ions from calcium carbonate minerals that are essential to the growth of the shells and skeletons of many marine organisms.