Intensity of Acidification
|
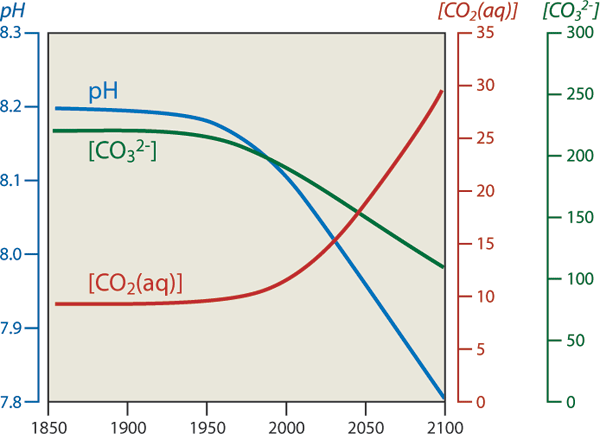
Whether it is nicknamed an “osteoporosis of the sea” or “climate change’s equally evil twin”, the speed of ocean acidification has doubtlessly opened the eyes of many scientists. Over the past 300 million years, ocean pH Earth’s oceans have maintained a relatively stable acidity level of approximately 8.2 on the pH scale. Then, only in the past 200 years we have decreased the pH level to 8.1. Although this decimal change may not seem significant, because the pH scale is logarithmic this represents an increase in acidity of almost 30 percent. This intense increase is partly because most of this CO2 is absorbed in the surface waters, rather than being diluted into the entire ocean. This causes an even more intense increase in acidification in a concentrated area.
The visible negative effects of this acidification on marine wildlife are staggering. Corals are bleaching, shellfish harvests are failing, and entire ecosystems are deteriorating. Although people still consider seawater to be relatively basic (neutral pH is 7), this increase threatens the survival of the marine organisms that have adapted over tens of millions of years to a relatively stable pH level. And oceans will continue to absorb the CO2 we emit today. Estimates of future CO2 levels, based on usual business emission levels, indicate that by the end of this century the pH of surface waters will decrease by another 0.5 units to pH 7.6, which is 150 percent more acidic than today. This pH level hasn't been experienced on Earth for over 20 million years. |
Estimated projections of ocean pH levels, CO2 levels and carbonate levels. |